Abstract
Introduction.
Minimal residual disease (MRD) is a powerful predictor of outcome in multiple myeloma (MM). A recent meta-analysis has confirmed this and demonstrated a hazard ratio for PFS of 0.41; 95% CI, 0.36-0.48; P < .001 (Munshi et al, JAMA Oncol, Jan 2017). We have previously demonstrated the prognostic impact of MRD both following ASCT in transplant-eligible (TE) patients and following induction in transplant non-eligible (TNE) patients. There is more limited data on the applicability and significance of MRD assessment in the maintenance setting, largely as a consequence of high rates of drop-off historically within myeloma trials but improved outcomes have seen larger numbers of participants with samples at later timepoints.
Patients and Methods.
This analysis aims to assess the impact of MRD on PFS amongst patients receiving maintenance or no further therapy in the NCRI Myeloma XI trial. In this study patients were randomised between thalidomide (CTD) and lenalidomide (RCD) based induction therapies. For patients with a sub-optimal response to initial therapy, induction was supplemented with sequenced bortezomib-based induction (CVD). Intensively treated patients then proceeded to an autologous transplant and then responding patients from both intensive and non-intensive arms were subsequently randomised to maintenance with lenalidomide monotherapy, lenalidomide and vorinostat or no further therapy. Bone marrow aspirates were obtained prior to maintenance randomisation (100 days post ASCT for TE and at the end of (sequenced-) induction treatment for TNE) and 6 months post maintenance randomisation. This analysis represents a subset of 389 patients (median age 63.5 years) with an informative post maintenance randomisation bone marrow aspirate. MRD was assessed using flow cytometry (sensitivity 0.004%) with a minimum of 500,000 cells evaluated with six- or eight-colour antibody combinations including CD138/CD38/CD45/CD19/CD56/CD27 in all cases and CD81/CD117 added latterly.
Results.
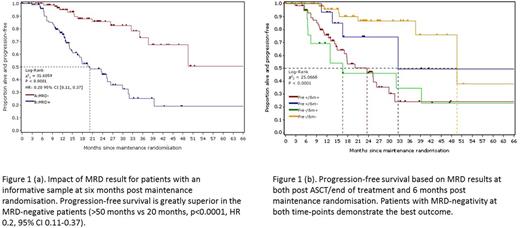
Taking the group as a whole, MRD-negativity was demonstrated in 206/389 (55.8%) and this was associated with a significant outcome advantage as the median PFS was >50 months versus 20 months for MRD-positive patients (Fig.1(a), p<0.0001, HR 0.2, 95% CI 0.11-0.37). When the pre-maintenance MRD result was also taken into account, outcome was best for patients achieving negativity post ASCT/end of treatment and remaining MRD-negative and worst for those patients who were MRD-positive post ASCT/end of treatment and remained so (Fig 1(b), p<0.0001). Conversions to MRD-negativity were seen in 32% of MRD-positive patients on maintenance compared to 4% of patients randomised to no further therapy (p=0.0045). This conversion is associated with some improvement in outcome, but this group still have inferior outcome relative to those patients achieving MRD-negativity earlier in protocol treatment. Conversions to MRD-positivity were also seen in 24 (9.5%) of 252 patients and the outcome for this patient group was similar to that of the patients who remain MRD-positive throughout (Fig. 1(b)). For those patients that remained MRD-positive, a benefit from maintenance could be demonstrated by a lower level of residual disease relative to those patients on observation (median level of neoplastic plasma cells 0.15% on maintenance vs 0.39%, p=0.04).
Conclusions.
We would conclude that MRD is a particularly powerful predictor of outcome in the maintenance setting and is clearly a desirable therapeutic goal in this patient group. The hazard ratio of 0.2 demonstrated here appears superior to those demonstrated in previous studies examining post induction or ASCT time-points. Approximately one third of MRD-positive patients receiving maintenance became MRD-negative and maintenance therapy also results in a decrease in disease levels in those patients remaining positive. These results support the role of MRD monitoring in assessment of the efficacy of different maintenance/consolidation strategies within clinical trials. In the longer term, a stratified approach to treatment based on sequential MRD assessments is feasible. The predictive ability of MRD during maintenance will be assessed with respect to overall survival when the primary endpoint matures in September 2017 and presented at the meeting.
Rawstron: BD biosciences: Patents & Royalties; Gilead: Research Funding; Janssen: Consultancy, Honoraria; AbbVie: Consultancy, Honoraria; Roche: Consultancy, Honoraria. Pawlyn: Celgene: Honoraria, Other: Travel support; Janssen: Other: Travel support; Takeda: Honoraria, Other: Travel support. Davies: Bristol-Myers: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Consultancy, Honoraria; Seattle Genetics: Consultancy, Honoraria. Jones: Celgene: Honoraria, Other: Travel Support, Research Funding. Kaiser: Amgen: Consultancy, Honoraria; Celgene: Consultancy, Honoraria, Research Funding; Janssen: Honoraria; Takeda: Consultancy; BMS: Consultancy, Other: Travel expenses; Chugai: Consultancy. Drayson: Abingdon Health: Equity Ownership, Membership on an entity's Board of Directors or advisory committees. Jenner: Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Support , Research Funding, Speakers Bureau; Takeda: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel Support, Research Funding, Speakers Bureau; Chugai: Membership on an entity's Board of Directors or advisory committees. Gregory: Janssen: Honoraria; Celgene: Consultancy, Honoraria. Jackson: Celgene: Honoraria; J&J: Honoraria; Amgen: Honoraria; Takeda: Honoraria; Chugai: Honoraria. Morgan: Celgene: Consultancy, Honoraria, Research Funding; Takeda: Consultancy, Honoraria; Bristol Myers: Consultancy, Honoraria. Owen: Takeda: Honoraria, Other: Travel Support; Janssen: Consultancy, Other: Travel support; Celgene: Consultancy, Honoraria, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal